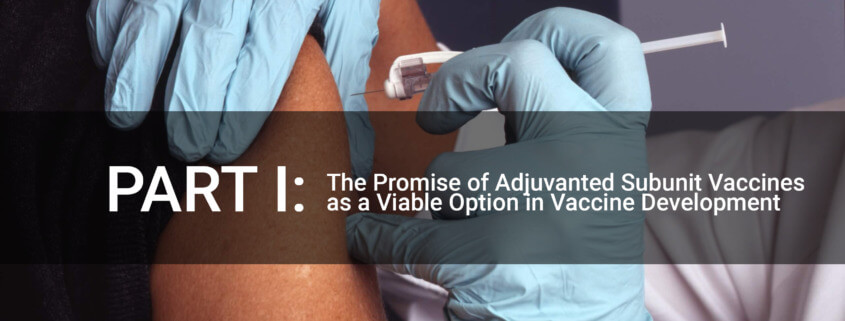
INTRODUCTION: Over the last several decades, subunit vaccines have shown consideration for vaccines. About 30 years ago, scientists developed a potential subunit vaccine for the varicella-zoster virus (VZV).1 A subunit vaccine’s advantage is several-fold, including not needing to grow and inactivate live viruses (or create an attenuated virus strain) and use the most antigenic portion of the virus antigen in question.
DISCUSSION: The key to this vaccine is that, unlike the live attenuated VZV vaccine, latency cannot be established when only a part of the virus protein (antigen) is used as the vaccine. In the subunit VZV vaccine’s initial stages, the VZV glycoprotein E (gE ) expresses in a recombinant vaccinia virus vector. Since regulatory agencies did not approve recombinant vaccinia vectors, scientists needed a different path. They constructed secretory truncated VZV glycoprotein gE with 511 amino acids into a vaccinia virus vector which would secret the glycoprotein. Next, they purified this from tissue culture fluids of the infected cells. The secreted gE protein-induced complement-dependent neutralizing antibodies in rabbits. Subsequent research showed that a VZV seropositive individual, when immunized with purified VZV gE in combination with an adjuvant, produced neutralizing antibodies.2 In the subsequent 20 years, GlaxoSmithKline’s clinical phase trials showed promising results with the truncated gE protein. As a result, the US Food and Drug Administration approved the VZV subunit vaccine (which used a GSK proprietary Adjuvant System, ASO1B), now called SHINGRIX, on Oct 20, 2017. Tavares-Da- Silva et al.3 found that based on the sale data from February 2019, with 9.4 million doses of vaccine, the efficacy (>90%) and the vaccine’s safety continue to mirror the clinical trials’ results. With the success of SHINGRIX, others have been encouraged to develop viral subunit vaccines. For example, respiratory syncytial virus (RSV) is a leading cause of acute lower respiratory tract infection in infants. Ensuing, scientists developed a recombinant RSV fusion glycoprotein (RSV F subunit). Leroux-Roels et al.4 found a single dose of the RSV subunit F vaccine was well-tolerated and enhanced pre-existing neutralizing antibodies through six months of follow-up. Awasthi, Hook, Shaw, and Friedman5 explain the development of a trivalent herpes simplex virus type 2 (HSV-2) vaccine using HSV-2 glycoproteins C, D, and E (gC2, gD2, gE2) using an alum adjuvant. This trivalent vaccine reduced the frequency of recurrent genital lesions and vaginal shedding of HSV-2 DNA by 50% (shedding of the replication-competent virus was almost eliminated). This result suggests the trivalent vaccine is a worthy candidate for immunotherapy of genital herpes. Furthermore, Tai et al.6 found that using a subunit vaccine containing the envelope protein of Zika is a strong candidate with high efficiency in preventing Zika virus infections in mice.
CONCLUSION: Subunit vaccines are being designed for many virus infections, based, in part, on the successful design and use of the VZV subunit vaccine, SHINGRIX. Only including the most immunogenic components of the virus antigenic protein in a vaccine offers many advantages, not the least of which is not needing to grow a live virus (whether an attenuated strain or a live virus to be inactivated before vaccination). Additional subunit vaccines will be appearing in the not-too-distant future.
REFERENCES:
- Vafai, A. (1993). Antigenicity of a candidate varicella-zoster virus glycoprotein subunit vaccine. Vaccine, 11(9) 937-940. https://doi.org/10.1016/0264-410x(93)90382-8
- Vafai, A. (1995). Boosting immune response with a candidate varicella-zoster virus glycoprotein subunit vaccine. Vaccine, 13(14), 1336-1338. https://doi.org/10.1016/0264-410x(94)00073-v
- Tavares-Da-Silva, F., Co, M. M., Dessart, C., Hervé, C., López-Fauqued, M., Mahaux, O., Van Holle, L., & Stemann, J.-U. (2019). Review of the initial post-marketing safety surveillance of the recombinant zoster vaccine. Vaccine, 38(18), 3489-3500. https://doi.org/10.1016/j.vaccine.2019.11.058
- Leroux-Roels, G., De Boever, F., Maes, C., Nguyen, T. L.-A., Baker, S., & Gonzalez, A. (2019). Safety and immunogenicity of a respiratory syncytial virus fusion glycoprotein F subunit vaccine in healthy adults: Results of a phase 1, randomized, observer-blind, controlled, dosage-escalation study. Vaccine, 37(20), 2694-2703. https://doi.org/10.1016/j.vaccine.2019.04.011
- Awasthi, S., Hook, L. M., Shaw, C. E., & Friedman, H. M. (2017). A trivalent subunit antigen glycoprotein vaccine as immunotherapy for genital herpes in the guinea pig genital infection model. Human Vaccines & Immunotherapeutics, 13(12), 2785-2793. https://doi.org/10.1080/21645515.2017.1323604
- Tai, W, Chen, J., Zhao, G., Geng, Q., He, L., Chen, Y., Zhou, Y., Li, F., and Du, L. (2019). Rational design of Zika virus subunit vaccine with enhanced. Journal of Virology, 93(17), e02187-18. https://doi.org/10.1128/JVI.02187-18
MKTG 1052 Rev A – 112420