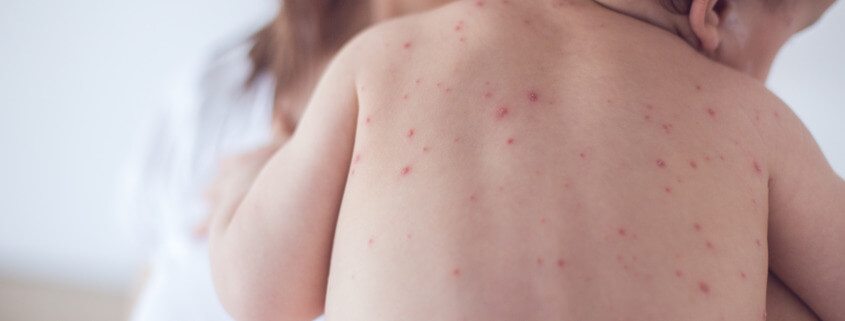
INTRODUCTION: Varicella-zoster virus (VZV) causes chickenpox, an acute viral, vesicular, exanthematous illness. After primary infection, VZV often becomes latent in ganglionic neurons, without production of viral proteins or infectious particles. In recent years, there have been numerous reports of children vaccinated with the live attenuated vaccine virus strain VZV vOka coming down with herpes zoster (HZ)1,2.
DISCUSSION: The effectiveness of the varicella vaccination program has been very good, with the occurrence of chickenpox virtually disappearing. However, more cases of viral reactivation leading to HZ have been seen in recent years among children vaccinated with vOka. The most serious complication in healthy children is the central nervous system infection. This is caused by reactivation of the vaccine virus from the dorsal root ganglia (DRG), usually years after the first varicella vaccination. Heusel and Grose1 looked at 12 cases of varicella vaccine meningitis.
The vOka vaccine strain was given to children in the United States for 26 years. It was approved in 1995 by the U.S. Food and Drug Administration as a single-dose regimen around age one year. Since 2012, Heusel and Grose have located eight cases of varicella vaccine meningitis in once-immunized children. Two of the eight children who received a single vaccine dose and developed varicella vaccine meningitis were immune-compromised. The other six children had no known immunosuppression. All of the children had HZ preceding onset of meningitis. However, there was an unusual aspect about the dermatomal distribution – usually a band of rash/blisters going around the waist.
Six of the eight zoster rashes were in a cervical dermatome (rash around the neck-shoulder-arm). Typical distribution of HZ following wild-type varicella include dermatomes T3-T10 and cranial nerve V (trigeminal ganglion). When the varicella vaccine was first approved in the U.S., there was a long catch-up period when older children could be immunized. It is most likely that several children in the study were immunized in the arm, rather than the thigh because they were older. Thus, they developed HZ in the cervical dermatomes. The current recommendation is for all children to have their first vaccine dose in the thigh around age one year.
A Medline literature search between 1960 and 2004 found only one case of HZ meningitis. Since 2017, four such cases in twice-immunized adolescents have been reported. In 2006, the FDA recommended a change from one to two doses with the first dose at age one and the second before entry to primary school (4-6 years). The belief was that varicella vaccine meningitis would not occur in children who had received two vaccine doses. This assumption has now proven to be incorrect. In contrast to children who had received only a single immunization, the time interval between first vaccination and the onset of varicella vaccine meningitis was constant; all four children were 14 years old.
The general pathogenesis is that following varicella vaccination in 50% of the children, the virus is carried within lymphocytes throughout the body. Some virus enters and becomes latent in the trigeminal ganglion, and when the virus reactivates, it can be carried via afferent fibers to the meninges, cerebral arteries, and eyes (herpes zoster ophthalmicus or HZO). Of the 12 published cases of HZ meningitis caused by the vaccine virus, only one case of HZO was seen.
CONCLUSION: The authors offered no consensus in explaining the varicella vaccine cases in immunized, immunocompetent children. They proposed several possible explanations:
(i) Waning immunity, as some military personnel who had received vaccination were 24% less likely to be VZV-seropositive than those recruits who had wild-type varicella as children;
(ii) Differential immune responses to vaccination vs. the varicella disease, perhaps due to quantitative differences in duration of their respective antibody responses (vaccine virus vs. wild-type virus). Here, a lowered antibody response to the major VZV structural glycoprotein (gC) was seen in immunized children vs. the VZV gC, which is a major component of wild-type VZV virions found abundantly in skin vesicles. During infection after vaccination, there is little or no formation of vesicles in the skin, thus much less VZV gC is produced; and
(iii) Emergence of wild-type alleles in the viral genome during reactivation of varicella vaccine virus as HZ.
The authors noted that in a case reported in 2019 of a severe HZ from the varicella vaccine virus, they observed the open reading frame (ORF) 0 had reverted to a wild-type allele2. Therefore, HZ was more severe in that child.
REFERENCES:
- Ethan H. Heusel and Charles Grose. (2020). Twelve Children with Varicella Vaccine Meningitis: Neuropathogenesis of Reactivated Live Attenuated Varicella Vaccine Virus. Viruses 2020, 12, 1078;1-13. https://doi.org/10.3390/v12101078
- Moodley, A., Swanson, J., Grose, C., and Bonthius, D.J. (2019). Severe Herpes Zoster Following Varicella Vaccination in Immunocompetent Young Children. Child Neurol. 34, 184-188. https://doi.org/10.1177/0883073818821498
By David Kilpatrick, PhD and Abbas Vafai, PhD
MKTG 1057 – Rev A 041821